Plasmapheresis, Intravenous Immunoglobin Show Promise as Early Treatment for COVID-19 - Pharmacy Times
Plasmapheresis, Intravenous Immunoglobin Show Promise as Early Treatment for COVID-19 - Pharmacy Times |
- Plasmapheresis, Intravenous Immunoglobin Show Promise as Early Treatment for COVID-19 - Pharmacy Times
- PlantForm signs agreement with Bio-Manguinhos/Fiocruz to develop a biosimilar pembrolizumab cancer drug - PRNewswire
- Merck Announces First-Quarter 2021 Financial Results - StreetInsider.com
| Posted: 29 Apr 2021 07:17 AM PDT 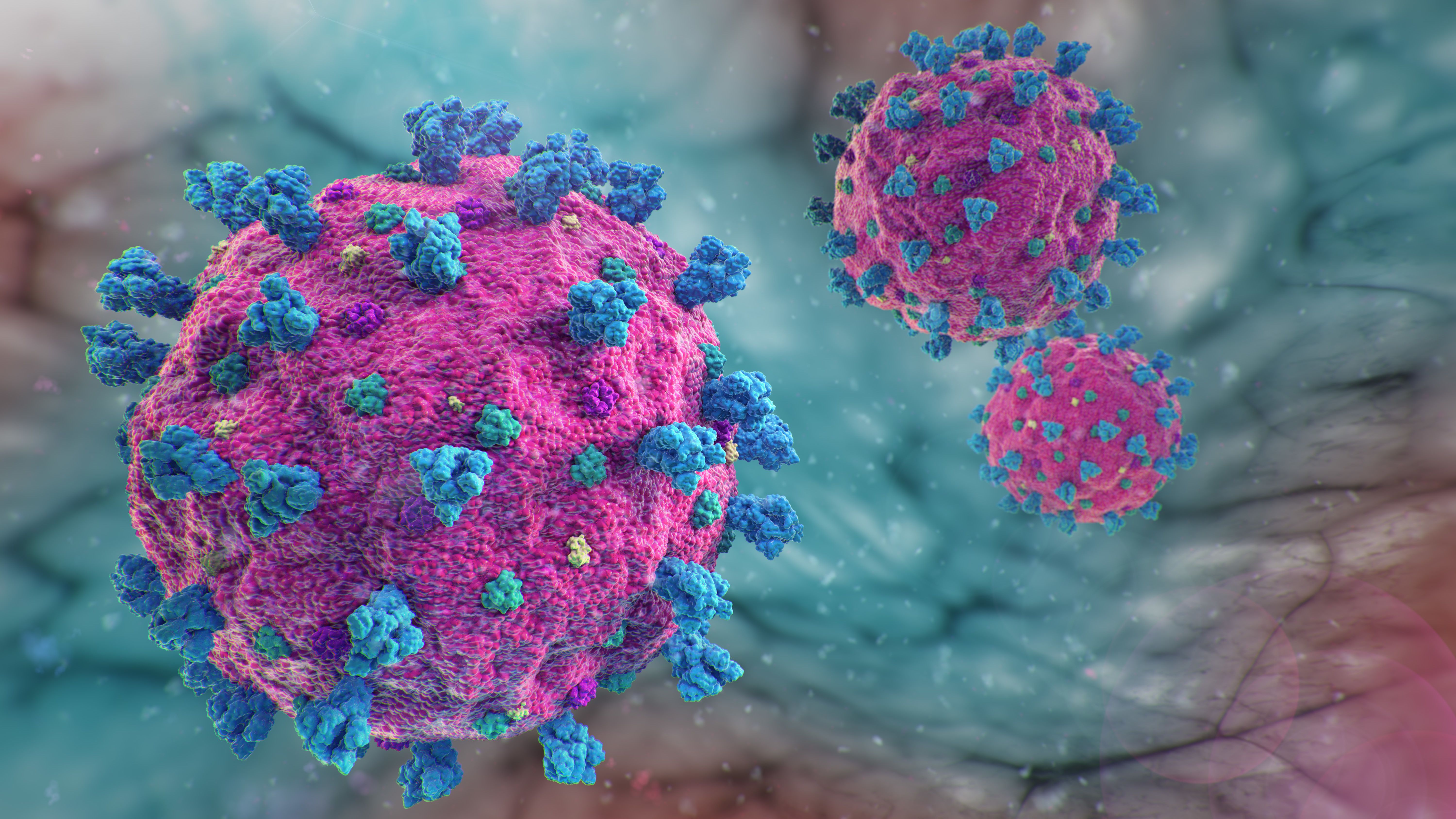 Plasmapheresis and intravenous immunoglobulin (IVIG) have been reported to be effective empirical therapeutic options to control COVID-19 infection. A metanalysis published in SN Comprehensive Clinical Medicine found that administering the therapies early in a patient's treatment could be associated with better outcomes. Plasmapheresis, the process of separating the plasma from blood cells, has been used in the treatment of multiple diseases, including myasthenia gravis, Guillain-Barre syndrome, and thrombotic microangiopathy. The process can remove a number of pathologic factors, including autoantibodies, complement products, lipoprotein, immune complexes, cryoglobulin, myeloma protein, ADAMTS-13, protein-bound toxins, cell platelets, and white blood cells. The process has limited adverse effects, including a fall in arterial blood pressure, arrhythmias, sensation of cold with elevated temperature, and paresthesia, all of which can be mitigated and responded to if the patient is closely monitored in a health care setting, according to the study. IVIG is a therapeutic choice for patients with antibody deficiencies and has been used in the treatment of a wide range of conditions, including heart failure, mycobacterial infection, adult respiratory distress syndrome, and Alzheimer disease. Currently, IVIG is used to stem the outbreak of viral diseases such as influenza, poliomyelitis, mumps, and measles. Initial reports have shown promising results for the use of IVIG in the treatment of COVID-19. Known complications are associated with other infectious diseases during transferring of blood substances or reaction to serum constituents, such as serum sickness, according to the study. Reports have shown that patients with COVID-19 admitted to the ICU have significantly higher levels of cytokines and chemokine in their blood. The cytokine storm, defined as a decrease in blood oxygenation, declined lymphocyte count over time, increased serum enzymes, elevated creatinine levels, and high levels of CRP, is understood to play a critical role in the pathophysiology of COVID-19 in critically ill patients. Several case reports have shown favorable results for using plasmapheresis and IVIG to prevent patients' conditions from worsening and recovering their lymphocyte count, indicating plasmapheresis and IVIG should be promptly administered to COVID-19 patients in order to have the highest efficacy in their treatment, according to the study. Clinical observation has shown that COVID-19 has 3 phases in symptomatic cases: a starting phase with subsequent viremia, an accelerating phase that is the vital phase of the infection, and a recovery phase with progressive lymphocytopenia and elevated inflammatory markers. Multiple studies have shown that administration of IVIG and plasmapheresis before day 14 of the illness could be associated with better outcomes, which may be due to the fact that viremia develops within the first week of infection, according to the study. As a result, the primary immune response first appears in the blood by day 10–14 and followed by viral clearance, according to the researchers. The study theorizes that convalescent plasma could be most effective if administered in the early stages of infection in order to minimize the patient's clinical deterioration. Research indicates that in COVID-19, IVIG can be used both prophylactically and in the treatment of the disease. The therapy has been shown to help prevent infection in individuals such as health care workers or patients who are at increased risk of disseminated infection. Further controlled clinical trials are being conducted to confirm the efficacy of using IVIG as a treatment for COVID-19. The investigators conclude plasmapheresis and IVIG are now the favorable options for prevention and treatment of COVID-19 cases that can be rapidly available and has low adverse effects and risks. REFERENCE: Ramtin Pourahmad, Bobak Moazzami, Nima Rezaei. Efficacy of Plasmapheresis and Immunoglobulin Replacement Therapy (IVIG) on Patients with COVID-19. SN Compr Clin Med. 2020 Jul; 31: 1–5. Accessed April 28, 2021. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7392618/ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Posted: 06 Apr 2021 12:00 AM PDT GUELPH, ON, and RIO DE JANEIRO, Brazil, April 6, 2021 /PRNewswire/ - PlantForm Corporation, PlantPraxis Biotecnologia and Bio-Manguinhos/Fiocruz (Health Ministry of Brazil) today announced a collaborative research and development agreement to develop a biosimilar pembrolizumab for the Brazilian market. The original antibody drug, pembrolizumab, is widely used in immunotherapy to treat a variety of cancers including melanoma, lung cancer, head and neck cancer, and stomach cancer. "This agreement advances PlantForm's collaborations in Brazil to make affordable, effective cancer drugs available to millions of Brazilians," said Dr. Don Stewart, PlantForm's President and CEO. "It also advances PlantForm's global strategy for biosimilar drug development and manufacturing to increase affordability and access to a wide range of life-saving medications." "Since 2015 when ANVISA approved the first biosimilar in Brazil it paved the way for more people to have access to advanced health treatments," said Germana Regazzi, PlantPraxis Director. "We want to make a difference in this market by transferring an innovative technology that will bring a low-cost biosimilar pembrolizumab to Brazil." "Bio-Manguinhos, a pioneer institution in the development and production of vaccines, biopharmaceuticals and diagnostics and a protagonist in the production of biosimilars in Brazil, is now investing in plant-based technology. We strongly believe it can be an important option to bring high quality/low cost biopharmaceuticals and vaccines to the Brazilian Universal Health System (SUS). PlantForm holds one of the most promising technologies in the field, representing an ideal partner to pursue this goal," commented Sotiris Missailidis, Deputy Director of Technological Development of Bio-Manguinhos/Fiocruz. The original drug (Keytruda® by Merck) is a global market leader in oncology treatment and is on track to be the world's best-selling drug by 2025 with annual projected sales of US $22.5 billion. It will lose exclusivity protection in the U.S., EU and Brazil in 2028. PlantPraxis is a joint venture between PlantForm do Brasil (a wholly owned subsidiary of PlantForm Corporation) and Axis Biotec Brazil to develop and commercialize certain biosimilar and/or biobetter versions of key biologic drugs using the vivoXPRESS® manufacturing platform for biopharmaceuticals. Bio-Manguinhos is a Brazilian government institution responsible for technology development and production of vaccines, reagents and biopharmaceuticals to meet public health needs. With this partnership, the three-year research and development initiative will culminate in protocols for Phase 1 and Phase 3 clinical trials. Following successful completion of clinical trials and market entry in Brazil through Bio-Manguinhos/Fiocruz, PlantForm and PlantPraxis will take biosimilar pembrolizumab to world markets. About PlantForm Corporation (www.plantformcorp.com) PlantForm Corporation is a biopharmaceutical company focused on the rapid development and production of biosimilar and specialty antibodies and proteins using the company's proprietary vivoXPRESS® platform. The plant-based vivoXPRESS® system makes it easier, faster and less expensive to produce biologics for approved and novel indications. PlantForm has an expanding portfolio of patents in seven families to protect both the core platform technology and products in development. About Bio-Manguinhos/Fiocruz (www.bio.fiocruz.br) As part of the Brazilian Ministry of Health, for 44 years the Institute of Technology on Immunobiologicals of the Oswaldo Cruz Foundation (Bio-Manguinhos/Fiocruz) has expanded access to health care for the population through the research, development and supply of products that prevent, diagnose and treat diseases. To establish itself as the largest public laboratory in Latin America, Bio-Manguinhos wrote parts of the History of Public Health in Brazil. It has played a decisive role in the eradication of poliomyelitis in the country and in the Americas; was instrumental in the control of measles and yellow fever, becoming one of the main producers of this vaccine in the world, having already exported to more than 70 countries. Partnerships with public and private institutions through technology transfer agreements, Partnerships for Productive Development (PDPs) and autochthonous development projects contribute to the expansion of its portfolio, which has more than 40 products among vaccines, in vitro diagnostics (IVDs) and biopharmaceuticals. Each year, millions of units are provided to the Brazilian Ministry of Health programs. For more information, please contact: Don Stewart Stacey Curry Gunn SOURCE PlantForm Corporation  | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Merck Announces First-Quarter 2021 Financial Results - StreetInsider.com Posted: 29 Apr 2021 03:37 AM PDT News and research before you hear about it on CNBC and others. Claim your 1-week free trial to StreetInsider Premium here.
KENILWORTH, N.J.--(BUSINESS WIRE)-- Merck (NYSE: MRK), known as MSD outside the United States and Canada, today announced financial results for the first quarter of 2021. This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20210429005381/en/ "While our results this quarter were impacted by the pandemic, the underlying demand for our innovative products remains strong and we remain confident in our future growth prospects," said Kenneth C. Frazier, chairman and CEO, Merck. "We are also taking the right steps to evolve Merck's operating model to continue to create value for patients, shareholders and society." "As I transition into the CEO role, one of my immediate priorities is to ensure that our experienced leadership team continues to build on our solid foundation," said Robert M. Davis, president, Merck. "Our company is well positioned for strong long-term performance, with scientific innovation remaining the source of our company's energy and value creation." Financial Summary
GAAP (generally accepted accounting principles) earnings per share assuming dilution (EPS) was $1.25 for the first quarter of 2021. Non-GAAP EPS of $1.40 for the first quarter of 2021 excludes acquisition- and divestiture-related costs, restructuring costs, income and losses from investments in equity securities and certain other items. Oncology Pipeline Highlights Merck continued to advance the development programs for KEYTRUDA (pembrolizumab), the company's anti-PD-1 therapy; Lynparza (olaparib), a PARP inhibitor being co-developed and co-commercialized with AstraZeneca; and Lenvima (lenvatinib mesylate), an orally available tyrosine kinase inhibitor being co-developed and co-commercialized with Eisai Co., Ltd. (Eisai), in addition to other notable developments as follows:
Business Development and Other Pipeline Highlights
Organon Highlights
Corporate Developments
First-Quarter Revenue Performance The following table reflects sales of the company's top pharmaceutical products, as well as sales of animal health products.
Pharmaceutical Revenue First-quarter pharmaceutical sales of $10.7 billion were in-line with the first quarter of 2020. Excluding the favorable effect of foreign exchange, sales declined by 3%. Sales performance reflects underlying strength in the business, offset by negative impacts of the COVID-19 pandemic, and the ongoing impacts of the loss of market exclusivity for several products. With respect to the COVID-19 pandemic, the estimated negative impact to Merck's first quarter pharmaceutical revenue was approximately $600 million. Continued reduced access to health care providers, combined with the prioritization of COVID-19 vaccines has negatively impacted the sales of certain products, notably vaccines in the United States. Pharmaceutical revenue reflects growth in oncology, largely driven by higher sales of KEYTRUDA, which rose 19% to $3.9 billion in the quarter, although the COVID-19 pandemic had a dampening effect on growing demand due to a decline in the number of new patients starting treatment. Global sales growth of KEYTRUDA reflects continued strong momentum from the non-small-cell lung cancer indications as well as continued uptake in other indications, including adjuvant melanoma, RCC, bladder, head and neck squamous cell carcinoma (HNSCC) and MSI-H cancers, as well as uptake following the recent launch of the 400mg every 6 weeks adult dosing regimen in the United States, partially offset by pricing pressure in Europe and Japan. Also contributing to growth in oncology was 57% growth in Lynparza alliance revenue, reflecting continued uptake in approved indications in the United States, Europe and China. The decline in vaccine sales was primarily driven by GARDASIL (Human Papillomavirus Quadrivalent [Types 6,11,16 and 18] Vaccine, Recombinant)/GARDASIL 9 (Human Papillomavirus 9-valent Vaccine, Recombinant), vaccines to prevent certain cancers and other diseases caused by HPV, primarily attributable to buying patterns in the United States and the timing of shipments in China, which in total negatively affected the year over year GARDASIL/GARDASIL 9 sales comparison by approximately $230 million. The COVID-19 pandemic also negatively affected sales for GARDASIL/GARDASIL 9, particularly in the United States and Europe. Also contributing to the decline in vaccine sales were lower sales of PNEUMOVAX 23 (pneumococcal vaccine polyvalent), a vaccine to help prevent pneumococcal disease, primarily reflecting the impact of the COVID-19 pandemic on demand in the United States, partially offset by higher volumes in international markets. Vaccines sales were also negatively affected by lower sales of ROTATEQ (Rotavirus Vaccine, Live Oral, Pentavalent), a vaccine to help protect against rotavirus gastroenteritis in infants and children, largely due to the timing of shipments in China and lower demand in the United States. Pharmaceutical sales in the quarter were negatively affected by the ongoing impacts from the loss of market exclusivity, including for ZETIA (ezetimibe) and NOXAFIL (posaconazole), as well as certain products in diversified brands. Performance in hospital acute care primarily reflects the decline in sales of ZERBAXA (ceftolozane and tazobactam) for injection, a combination cephalosporin antibacterial and beta-lactamase inhibitor for the treatment of adults with certain bacterial infections due to the temporary suspension of sales and product recall in the fourth quarter of 2020. Hospital acute care performance also reflects higher demand globally for BRIDION (sugammadex) Injection 100 mg/mL, a medicine for the reversal of neuromuscular blockade induced by rocuronium bromide or vecuronium bromide in adults undergoing surgery; and the continued uptake of PREVYMIS (letermovir), a medicine for prophylaxis (prevention) of cytomegalovirus (CMV) infection and disease in adult CMV-seropositive recipients of an allogeneic hematopoietic stem cell transplant. Animal Health Revenue Animal Health sales totaled $1.4 billion for the first quarter of 2021, an increase of 17% compared with the first quarter of 2020; excluding the favorable effect from foreign exchange, Animal Health sales grew 15%. Sales growth reflects higher demand globally for companion animal products, including parasiticide lines of products, primarily BRAVECTO (fluralaner), as well as higher sales of companion animal vaccines. Sales growth in livestock products reflects higher demand in international markets for ruminant, poultry and swine products, as well as higher demand globally for Animal Intelligence products. First-Quarter Expense, EPS and Related Information The tables below present selected expense information.
GAAP Expense, EPS and Related Information Gross margin was 69.6% for the first quarter of 2021 compared to 72.5% for the first quarter of 2020. The decrease reflects higher costs associated with COVID-19 development programs, including a charge related to the discontinuation of certain COVID-19 development programs, as well as higher acquisition- and divestiture-related costs, and pricing pressure, partially offset by favorable product mix. Selling, general and administrative expenses were $2.6 billion in the first quarter of 2021, an increase of 3% compared to the first quarter of 2020. The increase primarily reflects higher promotion and administrative costs, the unfavorable effects of foreign exchange and higher costs related to the company's planned spinoff of Organon, partially offset by lower selling costs due in part to the COVID-19 pandemic. Research and development expenses were $2.5 billion in the first quarter of 2021, an increase of 12% compared with the first quarter of 2020. The increase was primarily driven by higher expenses related to clinical development, including investment in COVID-19 development programs, as well as increased investment in discovery research and early drug development, partially offset by lower licensing costs. Other (income) expense, net, was $448 million of income in the first quarter of 2021 compared to $71 million of expense in the first quarter of 2020, primarily reflecting higher income from investments in equity securities in 2021 compared with 2020. The effective income tax rate of 8.0% for the first quarter of 2021 reflects a net tax benefit of $237 million related to the settlement of certain federal income tax matters. GAAP EPS was $1.25 for the first quarter of 2021 compared with $1.26 for the first quarter of 2020. Non-GAAP Expense, EPS and Related Information Non-GAAP gross margin was 75.7% for the first quarter of 2021 compared to 76.5% for the first quarter of 2020. The decrease in non-GAAP gross margin reflects higher costs associated with COVID-19 development programs, as well as pricing pressure, partially offset by favorable product mix. Non-GAAP selling, general and administrative expenses were $2.4 billion in the first quarter of 2021, an increase of 6% compared to the first quarter of 2020. The increase primarily reflects higher promotion and administrative costs and the unfavorable effects of foreign exchange, partially offset by lower selling costs due in part to the COVID-19 pandemic. Non-GAAP R&D expenses were $2.4 billion in the first quarter of 2021, a 13% increase compared to the first quarter of 2020. The increase primarily reflects higher expenses related to clinical development, including investment in COVID-19 development programs, as well as increased investment in discovery research and early drug development, partially offset by lower licensing costs. Non-GAAP other (income) expense, net, was $141 million of expense in the first quarter of 2021 compared to $169 million of expense in the first quarter of 2020. The non-GAAP effective income tax rate was 14.1% for the first quarter of 2021. Non-GAAP EPS was $1.40 for the first quarter of 2021 compared with $1.51 for the first quarter of 2020. A reconciliation of GAAP to non-GAAP net income and EPS is provided in the table that follows.
Financial Outlook The guidance provided below is based on the assumption that the Organon business will be part of Merck for all of 2021; however, the Company expects that the Organon spinoff will occur on June 2, 2021. If the spinoff occurs, these financial estimates will be updated. Initial information related to revenue from continuing operations is provided below. Merck continues to experience strong global underlying demand across its business. Consequently, at mid-April 2021 exchange rates, Merck continues to expect sales growth of 8% to 12% in 2021 with full-year 2021 revenue estimated to be between $51.8 billion and $53.8 billion, including a positive impact from foreign exchange of less than 2%. Merck now estimates that the pandemic will have a net unfavorable impact to 2021 revenues of approximately 3%, all of which relates to the pharmaceutical segment. Merck continues to believe that global health systems and patients have largely adapted to the impacts of COVID-19 disease, but that negative impacts will persist, particularly during the first half of 2021 and most notably with respect to vaccine sales in the United States, which is expected to be partially offset by the re-allocation of GARDASIL 9 doses to markets outside of the United States to address continued strong demand. Merck now expects full-year 2021 GAAP EPS to be between $5.05 and $5.25. Merck continues to expect full-year 2021 non-GAAP EPS to be between $6.48 and $6.68, including a positive impact from foreign exchange of less than 3%. The non-GAAP range excludes acquisition- and divestiture-related costs, costs related to restructuring programs, income and losses from investments in equity securities and certain other items. For full-year 2021, Merck expects the pandemic to have a negligible impact on operating expenses, as spending on the development of its COVID-19 antiviral programs is expected to largely offset the favorable impact of lower spending in other areas due to the COVID-19 pandemic. Neither the sales nor the EPS guidance ranges provided above include the impact of the potential launch of Merck's COVID-19 antiviral drug candidate. The following table summarizes the company's full-year 2021 financial guidance.
A reconciliation of anticipated 2021 GAAP EPS to non-GAAP EPS and the items excluded from non-GAAP EPS are provided in the table below.
Impact of Planned Spinoff of Organon Merck expects the spinoff of Organon to be completed on June 2, 2021. Merck continues to expect the transaction to create two companies with enhanced strategic and operational focus, improved agility, simplified operating models, optimized capital structures and improved financial profiles. Merck believes the transaction will deliver significant benefits for both Merck and Organon and create value for Merck shareholders. On a pro forma basis, assuming it operated as an independent company for the full year, Organon is expected to generate $6.1 billion to $6.4 billion in revenue in 2021. Organon is expected to have $9.5 billion in initial debt and is expected to pay a special tax-free dividend to Merck of approximately $9.0 billion. For Merck, the spinoff of Organon will allow it to increase its focus on key growth pillars, achieve higher revenue and EPS growth rates and enable incremental operating efficiencies of approximately $1.5 billion, which are expected to be achieved ratably over three years, with approximately $500 million realized during 2021. Merck will continue to incur overhead costs previously allocated to the Organon products, which are estimated to be approximately $400 million on a full-year basis. These costs are expected to be reduced over time and are netted into the overall efficiency target. Merck expects to use the special tax-free dividend from Organon for business development and/or share repurchases. As a result of the stronger growth Organon is expected to achieve as a standalone company and the benefit of operating efficiencies at Merck enabled by the spinoff, Merck expects combined non-GAAP EPS of the two companies to be higher within 12-24 months post-spinoff versus what would have been achieved assuming no transaction. Due to the higher relative profitability of Organon's products, Merck's operating margin from continuing operations is expected to initially be slightly lower in 2021 versus what it was prior to the spinoff. With the incremental operating efficiencies enabled by the spinoff, Merck's operating margins are expected to be higher within 12-24 months versus where they would have been in the absence of the spinoff and to be greater than 42% in 2024. Finally, assuming the completion of the Organon spinoff, Merck anticipates full-year 2021 revenue from continuing operations to be between $45.8 billion and $47.8 billion. Continuing operations for Merck exclude Organon results for the full year. Further details, including post-spinoff GAAP and non-GAAP EPS guidance, will be announced in conjunction with Merck's second-quarter 2021 earnings release. Earnings Conference Call Investors, journalists and the general public may access a live audio webcast of the call today at 8:00 a.m. EDT on Merck's website at https://investors.merck.com/events-and-presentations/default.aspx. Institutional investors and analysts can participate in the call by dialing (833) 353-0277 or (469) 886-1947 and using ID code number 7279283. Members of the media are invited to monitor the call by dialing (833) 353-0277 or (469) 886-1947 and using ID code number 7279283. Journalists who wish to ask questions are requested to contact a member of Merck's Media Relations team at the conclusion of the call. About Merck For 130 years, Merck, known as MSD outside of the United States and Canada, has been inventing for life, bringing forward medicines and vaccines for many of the world's most challenging diseases in pursuit of our mission to save and improve lives. We demonstrate our commitment to patients and population health by increasing access to health care through far-reaching policies, programs and partnerships. Today, Merck continues to be at the forefront of research to prevent and treat diseases that threaten people and animals – including cancer, infectious diseases such as HIV and Ebola, and emerging animal diseases – as we aspire to be the premier research-intensive biopharmaceutical company in the world. For more information, visit www.merck.com and connect with us on Twitter, Facebook, Instagram, YouTube and LinkedIn. Forward-Looking Statement of Merck & Co., Inc., Kenilworth, N.J., USA This news release of Merck & Co., Inc., Kenilworth, N.J., USA (the "company") includes "forward-looking statements" within the meaning of the safe harbor provisions of the U.S. Private Securities Litigation Reform Act of 1995. These statements are based upon the current beliefs and expectations of the company's management and are subject to significant risks and uncertainties. There can be no guarantees with respect to pipeline products that the products will receive the necessary regulatory approvals or that they will prove to be commercially successful. If underlying assumptions prove inaccurate or risks or uncertainties materialize, actual results may differ materially from those set forth in the forward-looking statements. Risks and uncertainties include but are not limited to, general industry conditions and competition; uncertainties as to the timing of the proposed spinoff; general economic factors, including interest rate and currency exchange rate fluctuations; the impact of the global outbreak of novel coronavirus disease (COVID-19); the impact of pharmaceutical industry regulation and health care legislation in the United States and internationally; global trends toward health care cost containment; technological advances, new products and patents attained by competitors; challenges inherent in new product development, including obtaining regulatory approval; the company's ability to accurately predict future market conditions; manufacturing difficulties or delays; financial instability of international economies and sovereign risk; dependence on the effectiveness of the company's patents and other protections for innovative products; and the exposure to litigation, including patent litigation, and/or regulatory actions. The company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise. Additional factors that could cause results to differ materially from those described in the forward-looking statements can be found in the company's 2020 Annual Report on Form 10-K and the company's other filings with the Securities and Exchange Commission (SEC) available at the SEC's Internet site (www.sec.gov).
View source version on businesswire.com: https://www.businesswire.com/news/home/20210429005381/en/ Media Contact: Patrick Ryan Investor Contacts: Peter Dannenbaum Raychel Kruper Source: Merck & Co., Inc. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| You are subscribed to email updates from "poliomyelitis treatment" - Google News. To stop receiving these emails, you may unsubscribe now. | Email delivery powered by Google |
| Google, 1600 Amphitheatre Parkway, Mountain View, CA 94043, United States | |

Comments
Post a Comment